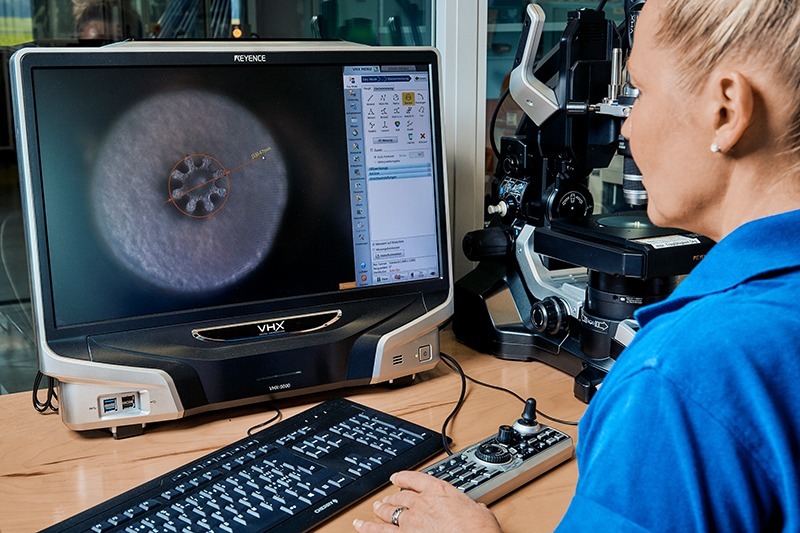
GENESIS Tech Center services
Work with Tegra Medical’s GENESIS Tech Center® team to help give your product a smooth launch to market. Our product development services include Design For Manufacturing (DFM), prototyping, pilot production, and more.
These product-launching services are the first step of our true end-to-end manufacturing solutions, working in concert with production, assembly, and packaging. From the “business end” to the handle and everything in between we tackle your entire project.
Refine your design
Do you have a great product design that needs some fine-tuning before it can be manufactured efficiently? Product design assistance, including DFM, reduces cost, increases quality and shortens the development cycle of your medical device.
Tegra Medical’s GENESIS Tech Center engineers work as a team with your engineers, allowing us to explore alternatives in processes and materials and immediately see the cost impact.
Learn how we can help make your production process as efficient and cost-effective as possible.
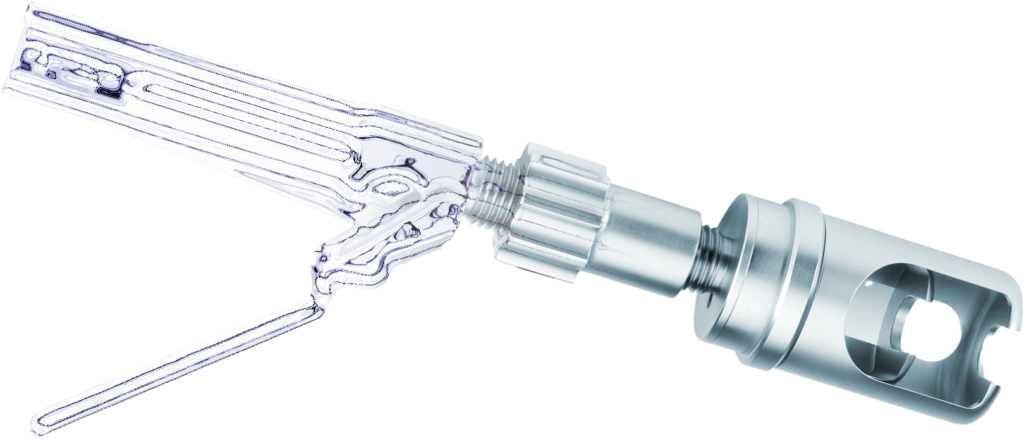

Prototyping
We manufacture your prototypes to spec so you can get them into the lab or clinical trials and ultimately to market sooner. All facilities use the same type or even the exact same pieces of manufacturing equipment for prototyping as our lean production environment.
Our prototyping expertise means that once your design is frozen and the manufacturing process has been established, the scale-up for your commercial launch is seamless.
Pilot production
Meet tough deadlines for product or process evaluations, one-off product builds, and prototypes.
Pilot-produce single-level components or finished assemblies. We offer complete documentation and traceability as required.
